Résumés
Abstract
Epilithon is a complex community of autotrophic and heterotrophic organisms that includes inert, organic and inorganic material and is attached to the surface of submersed rocks. Water samples collected in the Grand River (southwestern Ontario) in April 2011 showed that ammonium concentrations decreased downstream, whereas nitrate varied, largely dependent on weather conditions (concentrations of both chemical species were higher during winter). Epilithon δ15N-TN downstream from the Kitchener wastewater treatment plant oscillated between 0.4 to 23.2‰, and δ13C-TC around -27‰. The wastewater treatment plant effluent consisted of δ15N-NO3- between 12 and 16‰, with a decreasing trend as it traveled downstream; δ15N-NH4+ became enriched downstream (as high as 31‰). Average values for δ13C-DIC were -10.1‰ and δ13C-DOC -26.8‰. It is proposed that the nitrogen and carbon isotope composition of epilithon could be used as a short- or medium-term environmental archive, as it reflects in-stream processes, such as ammonia oxidation, in a river impacted by treated wastewater. The interpretation provided here was limited due to the ample range of events and potential sources, specifically when the nitrogen isotopic composition of nitrate and ammonium was similar. Epilithon is easily collected, processed and analysed and proved to be valuable tool to describe changes in river and stream geochemistry.
Keywords:
- Epilithon,
- isotope composition,
- wastewater,
- rivers,
- environmental archive
Résumé
L’épilithon est une communauté complexe d'organismes autotrophes et hétérotrophes, qui vit conjointement avec des matériaux organiques et inorganiques attachés à la surface de rochers submergés. Des échantillons d’eau collectés dans la rivière Grand (sud de l’Ontario) en avril 2011 ont montré une diminution des concentrations d'ammonium vers l’aval, alors que les concentrations de nitrate variaient, principalement en fonction des conditions météorologiques (les concentrations des deux espèces chimiques furent plus élevées pendant l'hiver). La composition isotopique de l’épilithon en aval de l’usine de traitement des eaux usées a varié entre 0,4 et 23,2 ‰ pour l’azote ((d15N-TN) alors que pour le carbone (d13C-TC) la valeur était autour de -27 ‰. L'effluent de l'usine de traitement des eaux usées montrait des valeurs de δ15N-NO3- entre 12 et 16 ‰, avec une tendance à la baisse vers l'aval; les valeurs pour δ15N-NH4+ avait une tendance à augmenter vers l’aval (aussi élevé que 31 ‰). Les valeurs moyennes de δ13C pour le carbone inorganique étaient de 10,1 ‰ et celles pour le carbone organique (δ13C-DOC) étaient de -26,8 ‰. On propose que la composition isotopique de l’épilithon puisse être utilisée comme archive de l'environnement à court ou à moyen terme, étant donné que l’épilithon reflète les processus ayant lieu en amont, comme l’oxydation de l'ammoniac, dans une rivière touchée par le rejet d’eaux usées. Donc, δ15N-TN et δ13C-TC pourraient être utilisés comme un indicateur environnemental à court terme pour les rivières touchées par l’activité humaine, comme constaté dans la rivière Grand. L’interprétation des données actuelles était limitée en raison de la grande gamme de sources potentielles, en particulier lorsque les compositions isotopiques de l’azote étaient similaires pour le nitrate et l’ammonium. L'épilithon est facile à recueillir, à traiter et à analyser et il s'est révélé un outil précieux pour décrire les changements géochimiques se produisant dans la rivière.
Mots-clés :
- épilithon,
- composition isotopique,
- eaux usées,
- rivières,
- indicateur environnemental
Corps de l’article
1. Introduction
Epilithon is the concurrent assemblage of autotrophic and heterotrophic organisms that colonize and develop attached communities on the surface of submersed rocks. It comprises several taxa such as algae, bacteria, fungi, protozoa and detritus, among the most important (AZIM et al., 2005). It is also sometimes called biofilm; however, this term is preferred in sewage treatment contexts, for instance, algae or bacteria that are immobilized or physically retained in order to improve the handling of organisms and enhance nitrogen removal from wastewater (ABE et al., 2003; GEBARA, 1999). Epilithon is formed by two main components: cell biomass and extracellular polymeric substances (EPS), the latter mainly composed of polysaccharides, with small amounts of proteins, organic acids and minerals (AZIM and ASAEDA, 2005).
The Grand River (south western Ontario, Canada) has been thoroughly characterized, with studies ranging from nutrient uptake (BARLOW-BUSCH et al., 2006), species composition along seasonal and nutrient gradients (METCALFE-SMITH et al., 2000; ROTT et al., 1998; SREENIVASA and DUTHIE, 1973), nitrogen cycling (ROSAMOND et al., 2011), fish ecology (COOKE and BUNT, 1999) to health risks from livestock rearing (DORNER et al., 2004) and human activities (LISSEMORE et al., 2006). The Grand River receives agricultural and urban inputs from both point (sewage plants, tile drainage) and non-point sources (agriculture, chiefly from manure spreading). Historically, the Grand River has experienced low oxygen concentrations during summer, due to the high organic load (MACCRIMMON and KELSO, 1970). Given the projected increase in population, the Region of Waterloo is currently upgrading two of its largest wastewater treatment plants (Waterloo and Kitchener). These upgrades will result in changes in nutrient loads and the quality of the effluent.
Preferential use of the 14N isotope has been reported for algae and macrophytes (NEEDOBA et al., 2003; JONES et al., 2004), due to enzymatic expression, temperature, physiological stress and substrate saturation (MARIOTTI et al., 1982; HANDLEY and RAVEN, 1992). Early research done by KOHL et al.. (1971) found that δ15N values of plants in streams that received runoff from agricultural land were similar to fertilizer-derived dissolved inorganic nitrogen. In other words, as chemical and biological reactions proceed, discrimination against 15N is noticeable because substrate is consumed and the pool of available nitrogen becomes depleted in the light isotope (CERNUSAK et al., 2009); therefore, it is possible to relate the isotopic composition of biomass in-stream with probable sources of nutrients. Additionally, plant taxa assimilate carbon through different metabolic pathways during photosynthesis; whereas bacteria carbon isotope composition would depend on carbon provenance; thus, it should be possible to estimate the contribution of different nutrient sources.
The purpose of this research is to provide evidence of the usefulness of epilithon as a tool to link the isotopic composition of epilithon with wastewater-derived nutrients, and to determine if it could be use as an environmental archive for short-term in-stream biogeochemical conditions and sewage impacts in the Central Grand River (Ontario, Canada).
2. Material and methods
2.1 Sample collection
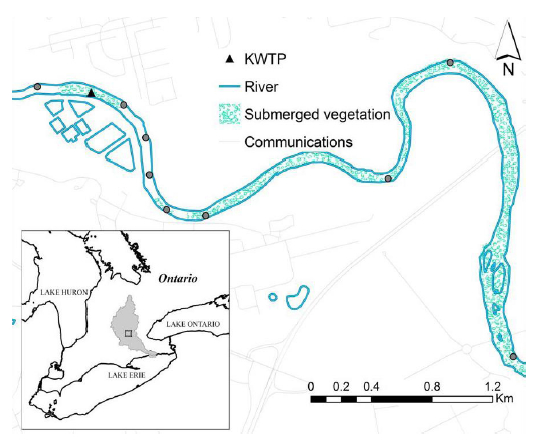
Epilithon was collected on April 19, 2011, along a 5700-metre reach in the central lower basin of the Grand River (Ontario), close to the Kitchener wastewater treatment plant (henceforth KWTP; Figure 1). The sampling was completed after snowmelt (water temperature > 10ºC), randomly choosing two small rocks of about 10 cm in diameter that were then stored in sealed plastic bags (Ziploc ®). A total of 10 sites were sampled, one location upstream and nine downstream from the KWTP outflow; samples were collected inside the plume developed by the treated effluent. The plume center was determined as those locations within the river with highest electrical conductivity. In the laboratory, all biological material was washed and scraped from the rocks with 20 mL of deionised water, using a stainless steel spatula and a soft nylon brush. The material was then centrifuged (2500 rpm, 15 min), the pellet was frozen and freeze-dried, and finally ground and stored in a dessicator for elemental (C and N) and isotopic analysis. The decanted supernatant was filtered through a quartz filter (1.2 μm pore size, Whatman Grade QM-A Quartz Filters) for elemental (C and N) and isotope analyses; no segregation by components was completed. It was assumed that the material retained by the filter represented macrogels and colloidal material (EPS), and might include picoplankton (0.2 - 2.0 μm) that did not pellet during centrifugation.
Figure 1
Sampling area, Central lower Grand River (south western Ontario) downstream from the Kitchener Wastewater Treatment Plant (KWTP). River flows south (left to right). Sampling locations are represented by circles; the dark triangle represents KWTP discharge.
Site d’étude, section centrale de la rivière Grand (le sud-ouest de l’Ontario) en aval de l’usine de traitement des eaux usées de Kitchener. La rivière coule vers le sud (de gauche à droite). Les sites d'échantillonnage sont représentés par des cercles; le triangle noir représente la décharge de l’usine de traitement des eaux usées.
2.2 Analysis
Biomass material was analysed before and after acidification (5% HCl) to address changes due to loss of carbonates. Extracellular polymeric substances (EPS) are considered here as the material collected on the 1.2 μm quartz filters. We acknowledge that this pore size would retain suspended solids and cells that did not pellet during centrifugation. However, the selection of pore size and material was dictated by the subsequent analytical procedures; only quartz filters could be used with the instrument mentioned below. Dry samples were analysed in a Delta Plus, Continuous Flow Stable Isotope Ratio Mass Spectrometer (Thermo Finnigan Bremen-Germany) coupled to a Carlo Erba Elemental Analyzer (CHNS-O EA1108 - Italy). Stable isotope ratios are expressed as delta (d) values and are measures of a ‘per mill’ (‰) relative to atmospheric air for nitrogen and the carbonate rock Peedee Belemnite for carbon. Errors are estimated to be ±0.3‰ for nitrogen and ±0.2‰ for carbon. Percentage compositions are calculated based on Carlo Erba Elemental Standards B2005, B2035 and B2036 with an error of ±1%. All samples were analysed in the Environmental Isotope Laboratory (EIL), University of Waterloo.
Dissolved inorganic nitrogen (DIN) was obtained by adding oxidized (NO2- + NO3-) and reduced (NH4+) species; the data shown represent spring and summer conditions. Water samples were filtered (0.45 μm pore size, Pall membrane filter), and stored cold or frozen before analysis. Nitrite and ammonium were measured by colorimetric methods; sulfanildamide for nitrite (BENDSCHNEIDER and ROBINSON, 1952) and indophenol for ammonium (SCHEINER, 1976), using a UV-VIS Beckman spectrophotometer. Nitrate measurement was performed on an ion chromatograph (Dionex Corp., USA). Samples for total phosphorus (TP, unfiltered samples) were acid digested in potassium persulfate solution for one hour, then assayed using the ascorbic acid - phosphomolybdate spectrophotometric method (Ultrospec 3100 spectrometer; MURPHY and RILEY, 1962). Statistical analyses (T-test) were performed with SPSS 13.0.
Values for δ15N-NO3- from water samples were obtained by chemical denitrification (NO3 to N2O) as described by MCILVIN and ALTABET (2005). Samples for δ15N-NH4+ were processed by an alkaline diffusion following SPOELSTRA et al. (2006); an alkaline solution (4 M KCl) is prepared so that a total volume of 20 mL (standard or sample) is placed in a 50 mL glass Teflon-lined jar, with an approximate mass of 15 μg of N. Diffusion traps were prepared by sealing acidified (10 μL 0.2 M H2SO4) quartz filter disks (Whatman QMA filters, baked at 550 ºC) in a section of polytetrafluoroethylene (PTFE) tape (‘T-Tape’), in order to allow complete diffusion of the NH3 onto the acidified disks. Each unit was adjusted to alkaline conditions (0.2 M NaOH) and buffered (pH ~9.3) with 2 mL of tetraborate buffer (0.1 M Na2B4O7 and 0.075 M NaOH). The jars were subjected to continuous shaking (80 rpm). After ten days the PTFE traps were removed and the filter disks retrieved and placed in 4.5 mL glass vials, frozen and then freeze dried. Isotopic analyses for nitrate and ammonia were determined using a GV Trace Gas preconcentrator systems coupled to a GVIsoprime mass spectrometer. Total Nitrogen isotope composition of biomass was determined with a Delta Plus, Continuous Flow Stable Isotope Ratio Mass Spectrometer (Thermo Finnigan/Bremen-Germany) coupled to a Carlo Erba Elemental Analyzer (CHNS-O EA1108 - Italy).
δ13C-DIC and δ13C-DOC data were provided by R.J. Elgood (unpublished data). δ13C-DIC was obtained from equilibrated headspace (He) after acidification (H3PO4 85%) to convert all dissolved inorganic carbon into CO2. The gas was analysed by Isochrome continuous flow GC-C-IRMS (Isotope Ratio Mass Spectrometer) and results referred to NBS19 (δ13C = 1.95‰). δ13C-DOC samples were stripped from CO2 and submitted to anaerobic oxidation of organic carbon (potassium persulfate) and analysed with an Isoprime Mass Spectrometer. Stable isotope ratios are expressed as delta (δ) values and are expressed of a ‘per mill’ (‰).
3. Results and discussion
3.1 Results
Within the study area, as the river flows downstream from KWTP, ammonium concentrations decreased, nitrate oscillated between 2.3 and 2.9 mg N•L‑1 (median), and dissolved inorganic nitrogen is largely driven by nitrate evolution, with a slight decreasing trend (Figure 2; data represent 2010 to 2012 spring and summer sampling events). Both ammonium (t = 2.98, α = 0.008) and nitrate (t = 4.99, α < 0.0001) were higher during cold months. Upstream δ15N-TN values from biomass and EPS were 7.8 and 8.3‰ respectively, whereas downstream values varied from 0.4 to 23.2‰ for biomass, being enriched further downstream (5700 m); EPS values were always below 8‰ (Table 1). Upstream EPS carbon isotope composition showed no noticeable differences after acidification, whereas epilithon δ13C showed a large shift, being as low as -31.6‰ (acidified sample) at 5700 metres below KWTP. The effluent comprised water enriched in δ15N-NO3- (12 - 16‰), with a decreasing trend as it traveled downstream; δ15N-NH4+ became enriched downstream (Figure 3). The average value for δ13C-DIC was -10.1‰ (±0.1, n = 4) and δ13C-DOC averaged -26.8‰ (±0.7; n = 3). Table 2 shows water chemistry (carbon, nitrogen and phosphorus) for samples collected in summer 2010 and spring 2011; the latter two are the main nutrients released into the river as a result of sewage treatment (Table 2, Figure 3).
Figure 2
Ammonium, nitrate and dissolved inorganic nitrogen concentrations (spring and summer 2010 to 2012) below the Kitchener (ON) wastewater treatment plant.
Concentrations de l’ammonium, de nitrate et d'azote inorganique dissous (printemps et été 2010-2012) en aval de l’usine de traitement des eaux usées de la ville de Kitchener (ON).
Table 1
Epilithon elemental analysis (C and N) and isotopic composition evaluated in Biomass and Extra Polymeric Substances (EPS) in a section of the Grand River (OM) impacted by wastewater effluent discharge.
L’analyse élémentaire de l’épilithon (C et N) et la composition isotopique mesurée dans la biomasse et les substances polymères extracellulaires (EPS) dans une section de la rivière Grand (ON) influencée par les effluents d’eaux usées.
Figure 3
δ15N values for water (NO3- and NH4+) and epilithon (TN). The dashed arrow represents the Kitchener (ON) Wastewater Treatment Plant.
Des valeurs de δ15N pour l’eau (NO3- et NH4+) et l’épilithon (TN). La ligne pointillée représente l’usine de traitement des eaux usées de la ville de Kitchener (ON).
3.2 Discussion
3.2.1 δ15N-TN
δ15N values are useful to identify potential sources and trophic levels (ANDERSON and CABANA, 2007; KOHZU et al., 2009) as well as to investigate the processes that yield specific signatures (COHEN et al, 2012). It is frequently observed that an increase in nutrient loads leads to an increase in biological uptake (DODDS et al, 2002). With enhanced nutrient availability, it is common to notice a shift in taxonomic composition, towards dominance by filamentous green algae and cyanobacteria, with a consequent increase in density and thickness of the epilithon layer (VERMAAT, 2005); such patterns have been observed in subsidized rivers (BOTHWELL, 1988; DAVIS et al, 1990). In-stream components and processes are affected by natural or man-made alterations. For instance, JONES et al. (2004) suggested that sediments are the best archive to record changes in lotic systems, yet epilithon becomes a very valuable tool to describe important changes when the river bed lacks a well-developed sediment layer. Taken together, its relative fast growth, its sessile condition and the ease with which it can be collected and analysed make epilithon a suitable candidate to be used as a monitoring tool in subsidized rivers.
SCOTT et al. (2008) reported that an increase in nutrient load favoured algal productivity, as evaluated in streams with a wide range of N and P concentrations, but also that productivity reaches a saturation point. Visual inspection of rocks in the Grand River showed heavy colonization and the presence of filamentous algae, but biomass was not quantified. In oligotrophic environments, algae rely on grazer-generated or bacterial-regenerated nutrients (SCOTT et al., 2008), therefore optimizing nutrient use, but also leading to a gradual enrichment of the heavy isotope (15N). Our findings agree with those of UDY and BUNN (2001), who found enriched values of δ15N (> 10‰) for aquatic species such as Spirogyra, Hydrilla sp and Vallisneria sp, which could be explained as enrichment resulting from intense nitrogen cycling, since values for soil nitrogen are similar, but also close to the low end of δ15N-NO3-. Biomass accrual has been reported to follow a non-linear increase due to reduced mass transfer velocity, clogged micro channels, detachment by high water velocity, grazing activity and accumulation of toxic substances (SABATER and ADMIRAAL, 2005, and references within).
KOHL et al. (1971) reported inorganic nitrogen with an isotopic signature of 7.8 ±0.57‰, the upper range for fertilizers, whereas total organic matter was 3.8 ±0.18‰, in agreement with expected values for soil organic nitrogen (Figure 5). δ15N-NH4+ is enriched downstream from the effluent as a result of oxidation, uptake and volatilization; therefore, it is possible that these three processes yield the particular epilithon isotope composition 5700 m downstream from KWTP. On the other hand, δ15N-NO3- values downstream showed a relatively stable trend (from 7 to 9‰) that may be due to nitrification uptake, as the former process would deplete it, whereas the latter would enrich it. Since the river upstream seems not to be nitrogen limited, the epilithic community might be using nitrate, the dominant dissolved inorganic nitrogen species. After the treatment plant discharges into the river, the epilithic community would be able to use not only all available ammonium (an energetically preferred form), but also nitrate; this could explain the large variability in δ15N-TN observed for epilithon as the plume travels downstream. When the plume has traveled more than 5000 m, the remnants of NH4+ in solution would have been taken up by epilithic biomass, as evidenced by the high values obtained (23.2‰; Figure 4). Although we do not provide information about inland sources of nutrients (soil runoff or fertilizers), we consider that the effect of the wastewater treatment plant is evident on the elevated δ15N-TN of epilithon 5000 m downstream from the KWTP effluent (relative to the samples collected between 0 and 1000 m), where enriched NH4+ (δ15N-NH4+ = 30.2‰) is taken up by this community. It is important to mention that uptake could vary with river discharge, where higher flow velocities, reduce the contact time of the bulk water with epilithon, which could also result in a lag period between biological activity and observed changes of the overlaying water column (ENSIGN and DOYLE, 2006). The above mentioned factors could also contribute to the large variability in the δ15N-TN values observed for the epilithon and potential dilution of the KWTP signature.
Figure 4
Epilithon nitrogen and carbon isotope composition (δ15N-TN and δ13C-TC) related to distance along the river. Note the differences in δ13C due to acidification.
Composition isotopique de l’azote et du carbone (δ15N-TN et δ13C-TC) de l’épilithon en fonction de la distance le long de la rivière. Notez les différences dans δ13C qui résultent de l'acidification.
It is also important to note that the river in this specific section has received discharges from another WTP, located upstream (Waterloo wastewater treatment plant). At a site 3000 m downstream from the Waterloo treatment plant, δ15N-NO3- was reported to be +7‰ (MURRAY 2008), similar to the upstream values for δ15N-TN obtained in the present study. Values reported for δ15N-NO3- in the reaches upstream of the two largest treatment plants in the region oscillate between +8.5‰ (winter) and +14‰ (summer; data not shown); the slight decrease could be interpreted as the result of the discharge of recently nitrified effluent into the river.
In relatively poor-nutrient environments (SIMON et al., 2003), epilithon values for δ15N were around 8‰, close to soil organic nitrogen values. Such values would be expected in the upstream section of the river (before the discharge of treated water), which could also be considered as the agricultural background isotopic composition. δ15N-NO3- values between 5 and 8‰ have been related to urban and agricultural impact to diverse extents (MAYER et al., 2003), yet the direct relationship of δ15N-NO3- values to epilithon is not completely clear. JONES et al. (2004) mentioned that more intense discrimination is present if nitrogen availability is low, since the internal cycling is important. Therefore, enriched values could be related to N-limited environments (or at least, lower than subsidized rivers or streams). At our study site, DIN differences upstream and downstream ranged from 0.5 mg N•L‑1 to 2.5 mg N•L‑1 (see Table 2), even though the daily ammonium discharge from the KWTP oscillates between 0.8 and 1.9 Ton N δ‑1 (≥ 20 mg N-NH4+•L‑1) due to changes in total discharge throughout the day. Also, it is important to state that the material collected could represent more than one growing season, therefore integrating and mixing different sources at the same time.
Tableau 2
Comparison of wastewater effluent and Grand River water chemistry upstream and downstream (500 m) from the Kitchener Wastewater Treatment Plant discharge. Data for July 2010 and April 2011 are shown as an example of summer and spring water chemistry.
Comparaison des effluents d’eaux usées et de la chimie de l’eau de la rivière Grand en amont et en aval (500 m) de l’usine de traitement des eaux usées de Kitchener. Les résultats obtenus en juillet 2010 et avril 2011 sont montrés comme un exemple de la chimie de l’eau pour l’été et le printemps.
Published literature has reported reduced discrimination against 15N with high nitrogen supply (YONEYAMA et al.,1991), but also evident discrimination at elevated external concentrations (CERNUSAK et al., 2009). It was expected that ammonium was the preferred species of nitrogen incorporated into biomass, given that is energetically favoured; thus, δ15N-TN would reflect ammonium incorporation, observed only at 5700 m downstream from KWTP (Figure 5a). However, a less selective nitrogen uptake could have happened in the Grand River below KWTP between 0 and 3600 m, given that both species could be assimilated into plant cells depending on external concentrations (DORTCH, 1990) or simultaneous transport across membranes (PRITCHARD and GUY, 2005). At this point, it is not possible to distinguish if the depleted nitrogen isotope composition in epilithon between 500 and 3600 m was the result of differential assimilation (e.g. discrimination) against nitrate or ammonium. An additional complication while interpreting the results is that nutrient regeneration has been identified as a process significantly affecting nutrient availability to cells growing within a polysaccharide matrix (SCOTT et al., 2008). A recent study by RIBOT et al. (2012) described a shorter plume in a highly seasonal river, where discharges from the wastewater treatment plant account for 100% of river discharge in summer, and the impact of the effluent was greater (δ15N-NH4+ up to 55‰) with an increasing trend in biomass nitrogen isotope composition as water travelled downstream (δ15N-TN ≈ 25‰ the highest value).
Figure 5
Nitrogen (a) and carbon (b) isotope composition for selected compartments; shaded rectangles represent values obtained in the Grand River (ON). 5a shows upstream and downstream differences in δ15N; 5b depicts differences in δ13C before and after acidification.
Composition isotopique de l'azote (a) et du carbone (b) pour les compartiments sélectionnés; les rectangles grisés représentent les valeurs obtenues dans la rivière Grand (ON). 5a montre les différences en amont et en aval pour δ15N; 5b illustre les différences pour δ13C avant et après acidification.
3.2.2 δ13C-TC
There was noticeable carbon depletion downstream from the wastewater effluent. This could be due to the mixing of allochthonous carbon in high order rivers such as the Grand River (-26‰.), also referred to by SCOTT et al. (2008), in which a large contribution of atmospheric CO2 (invasion) produces depleted in-stream δ13C (atmospheric δ13C-CO2 is -8.2‰). Different taxa will respond to inorganic carbon availability according to their different methods of carbon acquisition, yet a relation between the size of the watershed and the isotope composition of attached algae was found, with values of -23‰ in summer (FINLAY, 2004). According to the temporal variation, a more depleted isotopic composition in winter with respect to summer represents a lower dependence on atmospheric or respiration CO2, (autochthonous processes); therefore the gradual enrichment in open, canopied rivers could be interpreted as a dilution effect upon atmospheric CO2 incorporation. Abundant DOM, as a result of agricultural and KWTP inputs, together with carbonate incorporation from the water column, makes it difficult to interpret the δ13C data. Bicarbonate (HCO3-) use driven by CO2 limitation was not evaluated at this site, yet the DIC concentration (CO2+HCO3‑2) has been reported to be around 50 mg C•L‑1 in this section of the river (data not shown).
The heterotrophic bacterial community could incorporate DOC and POM from the water column; as a consequence their carbon isotope composition could be similar. VAN DEN MEERSCHER et al. (2009) showed depleted bacterial signatures, however they were similar to DOC and POC and clearly different from algae. This allowed them to propose that bacteria could be used as proxy for allochthonous carbon, whereas algae would represent autochthonous organic matter sources (Figure 5b). In our case, the epilithon is considered as the complete attached community, without taxa distinction. However, since algae represent CO2 or DIC incorporated in biomass, and this was released after acidification, is it possible that the depleted signature obtained in this study (≈ -27‰) represents heterotrophic bacteria. In addition, CRÉACH et al. (1997) found that bacterial isotopic fractionation was variable, depending upon DOC quality. Furthermore, δ13C values were similar to DOM isotope composition from the halophyte leachate used as a carbon source, which shows the allochthonous use of carbon by a heterotrophic bacterial community.
3.2.3 Future work
The potential and the actual role of attached periphyton in several settings has been described (BOWMAN et al., 2005; DAVIS et al., 1990; SOSIAK, 2002) relating the attached community to denitrification, but also to eutrophication alleviation by taking up the excess of nutrients. In most cases, bacterial and algal taxa are considered the attached assemblage (periphyton or epilithon). In the most heavily impacted section of the Grand River, the role epilithon plays in nitrogen cycling below wastewater treatment plant is being studied before and after wastewater treatment plant upgrading, and a better understanding of this site will give a deep insight into how different biotic compartments behave in temperate, subsidized rivers. There are some additional aspects to investigate, such as the intensity of the discrimination associated with different nitrogen species and the potential isotopic fractionation.
4. Conclusion
On the basis of the aforementioned results we suggest that epilithon could be used as a short-to-medium term environmental archive, for
it captures a given set of biogeochemical conditions in reasonable periods of time and the ease of collecting, processing and analysing samples. As the plume of nutrients travels downstream, the epilithon could also capture the dominant in-stream nitrogen processes, which was ammonia oxidation for this particular reach of the Grand River. It is also important to be aware of the ample range of event and mixed sources that could be recorded, thus complicating the correct interpretation of potential sources of nutrients and in-stream processes.
Parties annexes
Acknowledgement
The first author received a grant from the National Council of Science and Technology (CONACyT), Mexico (No 191667). This research was funded by an NSERC Strategic Grant. Special thanks to R. J. Elgood for his unconstrained and continuous support; W. Mark (EIL, University of Waterloo) for his help during the preparation, cleaning and analysis of samples. W.D. Taylor and an anonymous reviewer provided valuable comments; Grand River Conservation Authority (GRCA) provided GIS data from stream vegetation mapping; the Region of Waterloo provided water quality and discharge data, and granted permission to access the KWTP; landowners allowed access to the river at different locations. Special thanks to Peter Campbell (RSE-JWS Adjunct Scientific Director) for his comments and suggestions.
Bibliographical references
- Abe K., I. Matsumura, A. Imamaki and M. Hirano (2003). Removal of inorganic nitrogen sources from water by the algal biofilm of the aerial microalga Trentepohlia aurea. J. Microbiol. Biotech., 19, 325-328.
- Anderson C. and G. Cabana (2007). Estimating the trophic position of aquatic consumers in river food webs using stable nitrogen isotopes. J. N. Am. Benthol. Soc., 26, 273-285.
- Azim M.E. and T. Asaeda (2005). Periphyton Structure, Diversity and Colonization. In: Periphyton: Ecology, Exploitation and Management. Azim M.E., M.C.J Verdegem, A.A. van Dam and M. Beveridge (Editors). CAB International, U.K., Chap. 2, pp.15-34.
- Azim M.E., M.C. Beveridge, A.A. van Dam and M.C.J. Verdegem (2005). Periphyton and Aquatic production: an Introduction. In: Periphyton: Ecology, Exploitation and Management.Azim M.E., M.C.J Verdegem, A.A. van Dam and M. Beveridge (Editors). CAB International, U.K., Chap. 1, pp. 1-14.
- Barlow-Busch L., H.M. Baulch and W.D. Taylor (2006). Phosphate uptake by seston and epilithon in the Grand River, southern Ontario. Aquat. Sci., 68,181-192.
- Bendschneider K. and Robinson R.J. (1952). A new spectrophotometric method for the determination of nitrite in sea water. Technical Report No.8, University of Washington Oceanographic Laboratories, 18 p.
- Bothwell M.B. (1988). Growth rate responses of lotic periphytic diatoms to experimental phosphorus enrichment: the influence of temperature and light. Can. J. Fish. Aquat. Sci., 45, 261-270.
- Bowman M.F., P.A Chambers and D.W. Schindler (2005). Changes in stoichiometric constraints on epilithon and benthic macroinvertebrates in response to slight nutrient enrichment of mountain rivers. Freshwater Biol., 50, 1836-1852.
- Cernusak L. A., K. Winter and B. L Turner (2009). Plant δ15N correlates with the transpiration efficiency of nitrogen acquisition in tropical trees. Plant Physiol., 151, 1667-1676.
- Cohen M J., J.B. Heffernan, A. Albertin and J.B Martin (2012). Inference of riverine nitrogen processing from longitudinal and diel variation in dual nitrate isotopes. J. Geophys. Res., 117 G01021.
- Cooke S.J. and C.M. Bunt (1999). Spawning and reproductive biology of the Greater Redhorse, Moxostoma valenciennesi, in the Grand River, Ontario. Can. Field Nat., 113, 497-502.
- Créach V., G. Bertru and A. Mariotti (1997). Compositions isotopiques naturelles des bactéries hétérotrophes et détermination de l'origine du carbone organique dissous biodisponible. C.R. Acad. Sci. III-Vie., 320, 339-347.
- Davis L.S., J.P. Hoffman and P.W. Cook (1990). Production and nutrient accumulation by periphyton in a wastewater treatment facility. J. Phycol., 26, 617-623.
- Dodds W.K., A.J López, W.B. Bowden, S.Gregory, N.B Grimm, S.K. Hamilton, A.E. Hershey, E. Martí, W.H. McDowell, J.L. Meyer, D. Morrall, P.J. Mulholland, B.J. Peterson, J.L. Tank, H.M. Valett, J.R. Webster and W. Wollheim (2002). N uptake as a function of concentration in streams. J. N. Am. Benthol. Soc., 21, 206-220.
- Dorner S.M., P.M. Huck and R.M. Slawson (2004). Estimating potential environmental loadings of Cryptosporidium spp. and Campylobacter spp. from livestock in the Grand River Watershed, Ontario, Canada. Environ. Sci. Technol., 38, 3370-3380.
- Dortch Q. (1990). The interaction between ammonium and nitrate uptake in phytoplankton. Mar. Ecol. Prog. Ser., 61, 183-201.
- Ensign S.H. and M.W. Doyle (2006). Nutrient spiralling in streams and river networks. J. Geophys. Res., 111, 2156-2202.
- Finlay J.C. (2004). Patterns and controls of lotic algal stable carbon isotope ratios. Limnol. Oceanogr., 49, 850-861.
- Gebara F. (1999). Activated sludge biofilm wastewater treatment system. Water Res., 33, 230-238.
- Handley, L. and J. Raven (1992). The use of natural abundance of nitrogen isotopes in plant physiology and ecology. Plant Cell Environ., 15, 965-985.
- Jones R.I., L. King, M.M. Dent, C.S. Maberly and C.E. Gibson (2004). Nitrogen stable isotope ratios in surface sediments, epilithon and macrophytes from upland lakes with differing nutrient status. Freshwater Biol., 49, 382-291
- Kohl D.H., G. B. Shearer and B. Commoner (1971). Fertilizer nitrogen: contribution to nitrate in surface water in a corn belt watershed. Science, 174, 1331-1334.
- Kohzu A., I. Tayasu, C. Yoshimizu, A. Maruyama, Y. Kohmatsu, F. Hyodo, Y. Onoda, A. Igeta, K. Matsui, T. Nakano, E. Wada, T. Nagata and Y. Takemon (2009). Nitrogen-stable isotopic signatures of basal food items, primary consumers and omnivores in rivers with different levels of human impact. Ecol. Res., 24, 127-136.
- Lissemore L., C. Hao, P. Yang, P.K. Sibley, S. Mabury and K.S. Solomon (2006). An exposure assessment for selected pharmaceuticals within a watershed in Southern Ontario. Chemosphere, 64, 717-729.
- Mariotti A., F. Mariotti, M. Champigny, N. Amarger and A. Moyse (1982). Nitrogen isotope fractionation associated with nitrate reductase activity and uptake of NO3− by pearl millet. Plant Physiol., 69, 880-884.
- Mayer B., E.W. Boyer, C. Goodale, N.A. Jaworski, N. Van Breemen, R.B. Howarth, S. Seitzinger, G. Billen, K. Lajtha, K. Nadelhoffer, D. Van Dam, L. Hetling, M. Nosal and K. Paustian (2002). Sources of nitrate in rivers draining sixteen watersheds in the northeastern U.S.: Isotopic constraints. Biogeochemistry, 57-58, 171-197.
- MacCrimmon H.R. and J.R.M. Kelso (1970). Seasonal variation in selected nutrients of a river system. J. Fish. Res. Board Can., 27, 837-846.
- McIlvin M.R. and M.A Altabet (2005). Chemical conversion of nitrate and nitrite to nitrous oxide for nitrogen and oxygen isotopic analysis in freshwater and seawater. Anal. Chem., 77, 5589-5595.
- Metcalfe-Smith, J.L., G.L. Mackie, J. Di Maio and S.K. Staton (2000). Changes over time in the diversity and distribution of freshwater mussels (Unionidae) in the Grand River, Southwestern Ontario. J. Great Lakes Res., 26, 445-459.
- Murphy J. and Riley J.P. (1962). A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta, 27, 31-36.
- Murray M. (2008). Evaluating the isotopic fingerprint of wastewater treatment plant nitrogen and its evolution in the Grand River. BSc. Thesis, University of Waterloo, 45 p.
- Needoba J.A., N.A. Waser, P.J. Harrison and S.E. Calvert (2003) Nitrogen isotope fractionation in 12 species of marine phytoplankton during growth on nitrate. Mar. Ecol. Prog. Ser., 255, 81-91.
- Pritchard E. S. and R. D. Guy (2005). Nitrogen isotope discrimination in white spruce fed with low concentrations of ammonium and nitrate. Trees-Struct. Func., 19, 89-98.
- Regional Municipality of Waterloo (2011). Transportation and Environmental Services: Kitchener Wastewater Quality Data.
- Ribot M., E. Martí, D. von Schiller, F. Sabater, H. Daims and T.J. Battin (2012). Nitrogen processing and the role of epilithic biofilms downstream of a wastewater treatment plant. Freshwater Sci., 31, 1057-1069.
- Rosamond M.S., S. Thuss, S.L. Schiff and R.J. Elgood (2011) Coupled cycles of dissolved oxygen and nitrous oxide in rivers along a trophic gradient in Southern Ontario, Canada. J. Environ. Qual., 40, 256-270.
- Rott E., H.C. Duthie and E. Pipp (1998). Monitoring organic pollution and eutrophication in the Grand River, Ontario, by means of diatoms. Can. J. Fish. Aquat. Sci., 55, 1443-1453.
- Sabater S. and W. Admiraal (2005). Periphyton as Biological Indicators in Managed Aquatic Ecosystems. In: Periphyton: Ecology, Exploitation and Management.Azim M.E., M.C.J Verdegem, A.A. van Dam and M. Beveridge (Editors). CAB International, U.K., Chap. 9, pp.159-178.
- Scheiner D. (1976), Determination of ammonia and Kjeldahl nitrogen by indophenol method, Water Res., 10, 31-36.
- Scott J.T., J.A. Back, J.M. Taylor and R.S. King (2008). Does nutrient enrichment decouple algal-bacterial production in periphyton? J.N. Am. Benthol. Soc., 27, 332-344.
- Simon K.S., E.F. Benfield and S.A. Macko (2003). Food web structure and the role of epilithic biomass in cave streams. Ecology, 89, 2395-2406.
- Sosiak A. (2002). Long-term response of periphyton and macrophytes to reduced municipal nutrient loading to the Bow River (Alberta, Canada). Can. J. Fish. Aquat. Sci., 59, 987-1001.
- Spoelstra J., M. Murray and R.J Elgood (2006). A simplified diffusion method for delta15N analysis of NH4+. Environmental Geochemistry Lab Technical Procedure 20. Available from Department of Earth and Environmental Sciences, University of Waterloo.
- Sreenivasa M.R. and H.C. Duthie (1973). Diatom flora of the Grand River, Ontario, Canada. Hydrobiol., 42, 161-224.
- Udy J.W. and S.E. Bunn (2001). Elevated δ15N values in aquatic plants from cleared catchments: why? Mar. Freshwater Res., 52, 347-351.
- Vermaat J.E. (2005). Periphyton Dynamics and Influencing Factors. In: Periphyton: Ecology, Exploitation and Management. Azim M.E., M.C.J Verdegem, A.A. van Dam and M. Beveridge (Editors). CAB International, U.K., Chap. 3, pp. 35-50.
- Van den Meerscher K., P. Van Rijswijk, K Soetaert and J.J. Middelburg (2009). Autochthonous and allochthonous contributions to mesozooplankton diet in a tidal river and estuary: Integrating carbon isotope and fatty acid constraints. Limnol. Oceanogr., 54, 62-74.
- Yoneyama T., T. Omata, S. Nakata and J. Yazaki (1991). Fractionation of nitrogen isotopes during the uptake and assimilation of ammonia by plants. Plant Cell Physiol., 32, 1211-1217.
Liste des figures
Figure 1
Sampling area, Central lower Grand River (south western Ontario) downstream from the Kitchener Wastewater Treatment Plant (KWTP). River flows south (left to right). Sampling locations are represented by circles; the dark triangle represents KWTP discharge.
Site d’étude, section centrale de la rivière Grand (le sud-ouest de l’Ontario) en aval de l’usine de traitement des eaux usées de Kitchener. La rivière coule vers le sud (de gauche à droite). Les sites d'échantillonnage sont représentés par des cercles; le triangle noir représente la décharge de l’usine de traitement des eaux usées.
Figure 2
Ammonium, nitrate and dissolved inorganic nitrogen concentrations (spring and summer 2010 to 2012) below the Kitchener (ON) wastewater treatment plant.
Concentrations de l’ammonium, de nitrate et d'azote inorganique dissous (printemps et été 2010-2012) en aval de l’usine de traitement des eaux usées de la ville de Kitchener (ON).
Figure 3
δ15N values for water (NO3- and NH4+) and epilithon (TN). The dashed arrow represents the Kitchener (ON) Wastewater Treatment Plant.
Des valeurs de δ15N pour l’eau (NO3- et NH4+) et l’épilithon (TN). La ligne pointillée représente l’usine de traitement des eaux usées de la ville de Kitchener (ON).
Figure 4
Epilithon nitrogen and carbon isotope composition (δ15N-TN and δ13C-TC) related to distance along the river. Note the differences in δ13C due to acidification.
Composition isotopique de l’azote et du carbone (δ15N-TN et δ13C-TC) de l’épilithon en fonction de la distance le long de la rivière. Notez les différences dans δ13C qui résultent de l'acidification.
Figure 5
Nitrogen (a) and carbon (b) isotope composition for selected compartments; shaded rectangles represent values obtained in the Grand River (ON). 5a shows upstream and downstream differences in δ15N; 5b depicts differences in δ13C before and after acidification.
Composition isotopique de l'azote (a) et du carbone (b) pour les compartiments sélectionnés; les rectangles grisés représentent les valeurs obtenues dans la rivière Grand (ON). 5a montre les différences en amont et en aval pour δ15N; 5b illustre les différences pour δ13C avant et après acidification.
Liste des tableaux
Table 1
Epilithon elemental analysis (C and N) and isotopic composition evaluated in Biomass and Extra Polymeric Substances (EPS) in a section of the Grand River (OM) impacted by wastewater effluent discharge.
L’analyse élémentaire de l’épilithon (C et N) et la composition isotopique mesurée dans la biomasse et les substances polymères extracellulaires (EPS) dans une section de la rivière Grand (ON) influencée par les effluents d’eaux usées.
Tableau 2
Comparison of wastewater effluent and Grand River water chemistry upstream and downstream (500 m) from the Kitchener Wastewater Treatment Plant discharge. Data for July 2010 and April 2011 are shown as an example of summer and spring water chemistry.
Comparaison des effluents d’eaux usées et de la chimie de l’eau de la rivière Grand en amont et en aval (500 m) de l’usine de traitement des eaux usées de Kitchener. Les résultats obtenus en juillet 2010 et avril 2011 sont montrés comme un exemple de la chimie de l’eau pour l’été et le printemps.